This is the twelfth of 13 posts describing the impacts of marijuana’s rescheduling. An homage to Phish’s historic run at Madison Square Garden in the Summer of 2017, Budding Trends Baker’s Dozen will address how rescheduling affects various areas of the law and our daily lives. Enjoy the run.
The rollercoaster ride that is the American hemp industry has been fascinating to watch (though we recognize some on the ride may use a different word).
In 2018, Congress opened a door for hemp through the 2018 Farm Bill. While the origin story is still subject to intense debate, it seems most likely that the intention of the 2018 Farm Bill — although not thoughtfully expressed in the plain text — was to make way for industrial hemp. The plain language, however, created an opportunity for consumable hemp products, and the intoxicating hemp industry was born. It didn’t take long for consumable hemp products to catch on like wildfire. We saw the industry fill a void and serious consumer demand, particularly in states that did not have state medicinal or adult-use marijuana programs.
In many ways, though, we recognized that there were concerns that hemp operators weren’t subject to the same regulatory framework and barriers to entry (i.e., costs and burden) as those in the marijuana space. Given the lack of regulation, the opportunity for certain disreputable hemp operators to prey on consumers was also there. We expected to see more regulation. But I don’t think even the best of the best on the Psychic Friends Network could have predicted a law (the Agriculture, Rural Development, Food and Drug Administration, and Related Agency Appropriations Act of 2026 or the “Agriculture Bill”) would be passed in 2025 criminalizing most existing hemp-derived consumer products. But that’s where we are.
And now, another twist. The DEA issued its order moving state-licensed medical marijuana and FDA-approved medications containing marijuana from Schedule I to Schedule III.
So, what’s next for hemp?
First, a Refresher on Where Hemp Stands
Our resident professor has covered the mechanics of the hemp ban in detail. The short version: On November 12, 2025, Congress, as part of a continuing resolution to end the government shutdown, fundamentally rewrote the federal definition of hemp. The new law measures total THC — not just delta-9 THC on a dry weight basis — and caps finished hemp products at a maximum of 0.4 milligrams of total THC per container. That’s a sea change. Industry estimates suggest that provision alone would eliminate roughly 95% of products currently on the market.
To be clear, enforcement doesn’t kick in until November 12, 2026, and we’re still watching to see if relief for the hemp industry is coming.
What Rescheduling Does (and Doesn’t) Do for Hemp
So, is rescheduling the relief the industry (and perhaps consumers) was looking for? Not directly.
The DOJ’s final order rescheduling two classes of marijuana does not directly affect hemp. Hemp is not marijuana.
The rescheduling order covers “FDA-approved marijuana products” and “marijuana subject to a state medical marijuana license.” Products that are currently in the Farm Bill hemp gray area — delta-8, delta-10, THCA flower, and others — are in a murky space. Hemp was removed from the Controlled Substances Act by the 2018 Farm Bill, and that status was not changed by the rescheduling order. But, under the hemp ban that goes into effect in November 2026, many of those products would fall back under the definition of marijuana in the Controlled Substance Act once the new law takes effect. To keep it simple: The order moving certain defined subsets of marijuana from Schedule I to Schedule III has no direct legal effect on hemp’s classification.
But — and this is a significant but — rescheduling does affect the landscape in ways that will matter to hemp operators indirectly.
The bigger implication of rescheduling for hemp may be the signal it sends. The DOJ’s order speaks approvingly of the state medical marijuana program framework and endorses a consumer market for cannabinoids under appropriate regulatory oversight. That framing is consistent with the kind of argument the hemp industry needs to make: that regulated access to CBD and other hemp-derived products serves legitimate consumer needs, has an established safety record, and deserves a coherent federal framework — not a midnight ban passed through an appropriations bill.
The President’s Truth Social Post: A Signal or Just a Post?
On April 23 — the day before the DOJ formally announced the rescheduling order — President Trump posted on Truth Social calling on Congress to protect Americans’ access to full-spectrum CBD products.
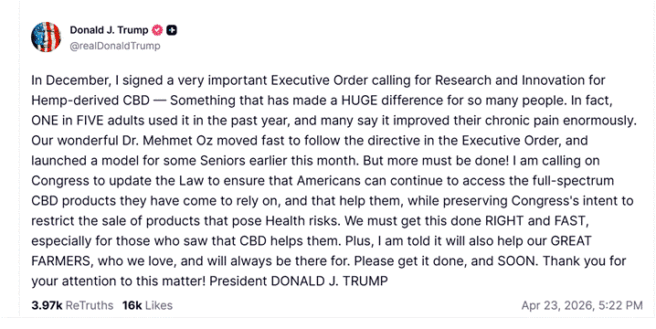
Trump cited his December executive order directing research and innovation on hemp-derived CBD, noted that Dr. Mehmet Oz at CMS had already launched a pilot program allowing some Medicare beneficiaries to access hemp-derived CBD products, and said plainly: “We must get this done RIGHT and FAST, especially for those who saw that CBD helps them.” He also nodded to America’s farmers, saying action on CBD would “help our GREAT FARMERS, who we love.”
This isn’t the first time the administration has signaled support for the hemp industry. Back in September 2025, Trump shared a video on Truth Social endorsing Medicare coverage of hemp-derived CBD, calling it potentially transformative for senior health. In December, he issued an executive order on hemp-derived CBD, directing the creation of research and regulatory pathways. And now the CMS pilot program has launched, with the FDA signaling it won’t interfere.
This is a pattern, not a one-off.
So, what does it mean? The honest answer: It’s complicated. The president signed the Agriculture Bill with the hemp ban provisions in it. He can express a preference for congressional action, but he can’t unilaterally undo the law he signed. What he can do is signal — loudly and publicly — what he wants Congress to do. And what he’s asking for seems to be targeted: preserve full-spectrum CBD while maintaining restrictions on products that pose genuine health risks. That’s a workable distinction, and it’s one hemp advocates have been pushing for all along.
What Hemp Operators Should Actually Be Watching
Given all of this, here’s where I’d focus attention:
The Farm Bill reauthorization is the most important legislative vehicle. The 2018 Farm Bill requires reauthorization by September 30, 2026 — before the hemp product ban takes effect in November. That is the most realistic opportunity to modify the Agriculture Bill’s hemp provisions. Hemp advocates are pushing to attach amendments that would revert the overly broad product restrictions while keeping the ban on synthetic cannabinoids and clearly intoxicating products. Advocacy in this process is likely essential to success.
Other congressional fixes are also in play. Several bills have already been introduced to delay or modify the November ban, including the Hemp Planting Predictability Act, which would delay enforcement by two years. Sen. Rand Paul has been a consistent and vocal critic of the ban. The president’s Truth Social post creates political momentum for a fix, particularly in farm-state congressional districts where hemp is an economic issue.
The CMS pilot program and the FDA’s posture matter. The administration has already implemented a model for Medicare coverage of hemp-derived CBD, with up to $500 in annual coverage for eligible beneficiaries. The FDA has signaled it won’t interfere with that program. This creates a real-world precedent for regulated CBD access that legislators can point to as a model. Operators should be monitoring that program closely and documenting its impact.
The June 29 rescheduling hearing has indirect relevance. The DOJ announced a new hearing beginning June 29 to consider whether marijuana broadly — including adult-use — should be Schedule III. That process will generate a record about the therapeutic benefits and appropriate regulatory frameworks for cannabinoids. Arguments made in that hearing about consumer access, safety, and regulatory structures will be relevant to the CBD policy debate as well.
The Bigger Picture
As a consumer, and not just a lawyer, I’ve seen how popular consumable hemp products have become. And as a lawyer who works in the cannabis space, I’ve fielded lots of questions at cocktail parties about whether my friends’ favorite products will really become a Schedule I substance in November. Last month, that answer was easier — yes, unless Congress provided some relief before November, most likely in the way of a new farm bill. But now the answer is more complicated and I’m, just perhaps, a little bit more optimistic. Rescheduling is a signal that the federal government is capable of moving on cannabis policy in a meaningful direction. The president’s Truth Social post is a signal that the White House understands the CBD issue and wants a solution. The CMS pilot program is a signal that the administration is willing to act administratively where it can. None of that saves the hemp industry on its own. November is still November, and the law is still the law until Congress changes it. But the stars are aligning in a way that makes a legislative fix more plausible than it looked a few months ago.
